What is the goal of confocal microscopy?
Confocal microscopy ties to obtain a specific, durable and strong signal (perhaps several signals) without degradation and respecting as much as possible the cellular functions.
What is Quantitative Confocal Microscopy?
The efficiency of the process must be constant over time and globally homogeneous in space for microscopy to be quantitative.
Fluorescent probes mark fixed cells, but also living cells.
What type of information can give us Confocal Microscopy?
– labeling of subcellular structures which generally correspond to
macromolecules or to a set of constituents (DNA, cytoskeleton proteins, …) or to constituent molecules of smaller size but present in significant concentration on an organelle
– coupled to an antibody, fluorochromes are also used as a developer of
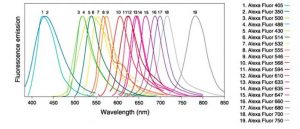
immunological reactions. Strong innovations are the development of supposed markers brighter and more stable, with narrower excitation and emission spectra (range of Alexa Fluors for example for immunocytochemical observations on fixed cells)
– measurement of rapid metabolic or physiological changes in cells
live (enzymatic activities, ionic movements, membrane permeability, apoptosis)
– study of protein dynamics and intra and intracellular movements in vivo.
Many markers for living cells can be used simultaneously:
(mitochondria excited and emitting in various parts of the spectrum, or various derivatives of Fluorescent Proteins
What is the he principle of fluorescence, the properties of probes
fluorescent and the influence of the environment? All these elements will have to be taken into account for answer the question: “What choice of fluorochromes? “.
Useful references:
fluorescence and spectroscopy Lakowicz (2006), Value (2002, 2004), on fluorochromes for Biology and Dyes: The Molecular Probes Handbook (anonymous, 2010) and Conn’s 10th edition (Horobin and Kiernan 2002).
What is the Principle of fluorescence?
Fluorescence is a form of luminescence that occurs as a result of photon absorption by a molecule of the fluorophore, fluorochrome or fluorescent probe type. It’s a phenomenon physics classified in the set of luminescence phenomena including the
photoluminescence (fluorescence, phosphorescence) and other types of luminescence (chemiluminescence, bioluminescence …). Photoluminescence is a radiative phenomenon following a bright excitation while chemiluminescence is a radiative phenomenon following a chemical reaction (true chemiluminescence) or enzymatic reaction (bioluminescence).
These different phenomena are essentially distinguished by the nature of the excitation energy involved.
LUMINESCENCE
PHOTOLUMINESCENCE CHEMILUMINESCENCE
BIOLUMINESCENCE
Fluorescence Phosphorescence
THERMOLUMINESCENCE
ELECTROLUMINESCENCE,
What is Photoluminescence?
Photoluminescence results in the emission of photons by a molecule that has been irradiated bya light beam generally in a range of wavelengths ranging from visible to
ultraviolet. Photoluminescence encompasses two processes: fluorescence andphosphorescence, which depend on the nature of the fundamental and excited states of the molecule Spencer BROWN & Christel POUJOL CNRS
Confocal Microscopy
There are different energy levels of a molecule and the different phenomena related to the photoluminescence.
The fluorescence takes place following a light absorption, it is characterized by the emission of photons that occurs during electronic transitions of a molecule between an excited state singlet S1 to ground state singlet S0 (relaxation). The bright excitation can be induced by a single photon (single-photon absorption) or by the simultaneous interaction of two (or three) photons whose energy sum is equal to an exciter photon to form an exciton (nonlinear optics: two-photon absorption, triphoton). For example two photons at ~ 700
nm can thus excite the DAPI as a single photon at 350 nm. (This principle is implemented in two-photon microscopy.) The time required for absorption is immediate, of the order of 10-15 sec (fs). The average life of the excited state is of the order of 10-9 to 10-7 s (> ns). Phosporescence is a type of phospholuminescence that persists after the cessation of excitation bright (as on a CRT screen). Indeed, it is distinguished from fluorescence by the mode of deexcitation of the molecule. Phosphorescence is characterized by transition.
